
by John Galbraith
Quinolones are a class of antibiotic with broad-spectrum antibacterial activity. They are well absorbed orally and have good tissue penetration making them popular agents for the treatment of a wide variety of infections. They are synthetic agents rather than natural products. The first member of the class, nalidixic acid, was discovered during the synthesis and purification of chloroquine in 1962. (1) The clinical role of nalidixic acid has been limited by its minimal serum levels and its narrow gram negative spectrum.
The core structure of the quinolone nucleus is depicted below: (2)
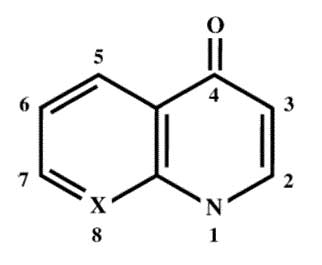
Second generation agents were developed by the addition of a fluoride atom at position 6 of the quinolone molecule, creating fluoroquinolones (FQ) with broadened antimicrobial spectrum including Pseudomonas species and some gram positive organisms. Fluoroquinolones were developed in the 1980s and included norfloxacin, ofloxacin, and ciprofloxacin. Further modifications in the 1990s led to the development of third generation FQ agents: levofloxacin and moxifloxacin, with improved Streptococcus pneumoniae coverage and fourth generation agents: trovafloxacin, with enhanced anaerobic activity. (2,3)



